AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Elog pilot logbook2/12/2023   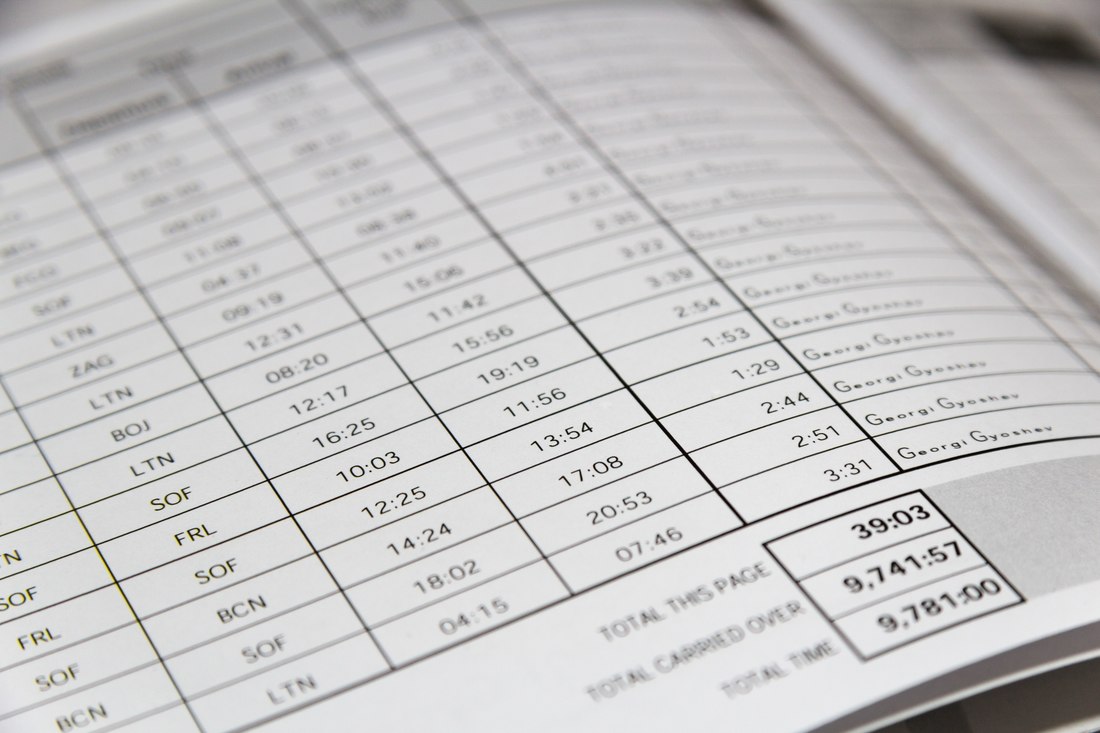
This solution also complies with other regulatory standards such as EU Annex 11, MHRA, GAMP, Good Manufacturing Practice (GMP), Standard Operating Procedures (SOP’s), and ISO. Additionally, the logbook software guarantees that details or logs are entered accurately and on time, and that they may be validated, reviewed, and authorized via approval workflows.ĪmpleLogic Equipment Usage Logbook Software is compliant with FDA regulations governing electronic records and signatures, including 21 CFR Part 11.  
This eLogbook Software is designed to assist users in converting manual paper forms to electronic equivalents. Electronic Logbook (eLogbook) Software for PharmaceuticalsĪmpleLogic Electronic Logbook is a web-based software or platform that records general production requirements and keeps track of Area and Equipment operational usage, Packing, Cleaning, Break Down, Clearance, and Preventive Maintenance, Fogging and Defogging Logs, Granulation, Calibration, Equipment Usage, Stability Schedule, Standards Usage, Service Logs, Dispensing, Production, Chemical Usage Logs, and many other equipment details log. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
